Diagnosis of rabies
Even with signs of the disease quite characteristic for rabies, like changes in behaviour or difficulties in swallowing the clinical observation and examination cannot rule out rabies nor confirm the diagnosis and hence, may only lead to a suspicion of rabies. Detection of the virus or some of its specific components using WHO and OIE recommended standard laboratory tests is therefore, the only way to undertake a reliable diagnosis of rabies.
Brain tissue is the preferred specimen for post-mortem diagnosis in both humans and animals. While intra-vitam diagnosis of rabies in animals is strongly discouraged, it is the only way of confirming infection in a patient suspected of having the disease. Intra-vitam diagnosis in suspect human patients is based on detecting virus or viral RNA in saliva, in innervated skin biopsy samples or hair follicles. However, due to intermittent shedding of virus in saliva and variable diagnostic sensitivity of the methods applied, only positive results are valid.
Detection of rabies antigen
 The most widely used primary diagnostic test for rabies in animals and humans fluorescent antibody test (FAT). This test is based on antigen detection and is recommended by both WHO and OIE as the gold standard for rabies diagnosis. Impression smears prepared from a composite sample of brain tissue are treated with antirabies serum or globulin labelled with fluorescein isothiocyanate (FITC). Specific aggregates of rabies virus antigen are detected by their fluorescence using a reflected light (incident light) fluorescence microscope. The FAT is accurate, sensitive and rapid, i.e. results can often be obtained within 1 to 2 hours of receipt of the specimen. Alternatively, particularly where fluorescence microscopy is not available, a direct rapid immunohistochemistry test (dRIT) was developed with a similar sensitivity and specificity as the FAT. The principle is similar to the FAT except that the dRIT uses streptavidin–biotin peroxidase staining.
The most widely used primary diagnostic test for rabies in animals and humans fluorescent antibody test (FAT). This test is based on antigen detection and is recommended by both WHO and OIE as the gold standard for rabies diagnosis. Impression smears prepared from a composite sample of brain tissue are treated with antirabies serum or globulin labelled with fluorescein isothiocyanate (FITC). Specific aggregates of rabies virus antigen are detected by their fluorescence using a reflected light (incident light) fluorescence microscope. The FAT is accurate, sensitive and rapid, i.e. results can often be obtained within 1 to 2 hours of receipt of the specimen. Alternatively, particularly where fluorescence microscopy is not available, a direct rapid immunohistochemistry test (dRIT) was developed with a similar sensitivity and specificity as the FAT. The principle is similar to the FAT except that the dRIT uses streptavidin–biotin peroxidase staining.
Lateral flow devices (LFDs) for rapid detection of rabies virus antigen under field conditions have been developed, however only partly meeting expectations in terms of test characteristics (sensitivity and specificity). Adequate validation according to international standards is still required so that they can be useful in surveillance situations in which the resources for the use of recommended testing are lacking.
Detection of rabies virus replication: inoculation tests
The other group of available techniques aim at detecting the replication of the virus on living substrates, e.g. cells. Virus isolation may be necessary to confirm inconclusive results in FAT/dRIT and for characterization of the virus strain. In neuroblastoma cells, rabies virus grows generally without cytopathic effect; once again it is necessary to use FAT to confirm the presence of rabies virus. After intracranial application, rabies induces clinical signs in mice that are relatively typical but have to be confirmed by FAT. Since cell culture is as sensitive as the mouse inoculation test, units should be established in laboratories to replace mouse inoculation tests as it avoids the use of life animals, is less expensive and gives more rapid results.
Detection of rabies virus RNA
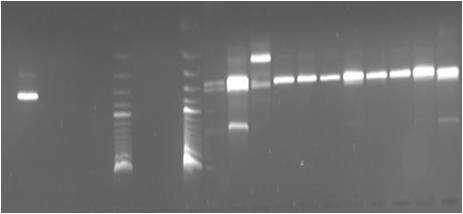 The reverse transcriptase (RT) polymerase chain reaction (PCR) is used to amplify a certain fragment of the virus genome (viral RNA). More recently, real-time PCR has been developed to increase sensitivity and to obtain results even faster. Those techniques have the highest level of sensitivity
The reverse transcriptase (RT) polymerase chain reaction (PCR) is used to amplify a certain fragment of the virus genome (viral RNA). More recently, real-time PCR has been developed to increase sensitivity and to obtain results even faster. Those techniques have the highest level of sensitivity
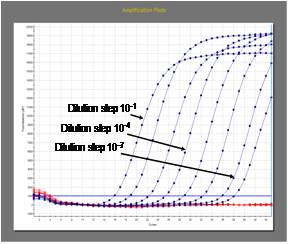 RT-PCR may be used as a confirmatory test and is particularly useful for intra vitam diagnosis in humans. Standardization and very stringent quality control is required. As PCR can produce false positive or false negative results it should only be used in combination with other conventional techniques.
RT-PCR may be used as a confirmatory test and is particularly useful for intra vitam diagnosis in humans. Standardization and very stringent quality control is required. As PCR can produce false positive or false negative results it should only be used in combination with other conventional techniques.
Serological tests
 Serological assays are not suitable for diagnosis of rabies infections in humans and animals as virus-specific antibodies in serum tend to appear only relatively late after the onset of clinical signs if at all. They are mainly used to evaluate the immune response to human and animal rabies vaccines.
Serological assays are not suitable for diagnosis of rabies infections in humans and animals as virus-specific antibodies in serum tend to appear only relatively late after the onset of clinical signs if at all. They are mainly used to evaluate the immune response to human and animal rabies vaccines.
The gold standard is the virus neutralisation test. Virus neutralising antibody titres directly correspond to the level of protection. Virus neutralisation assays are also the prescribed tests for international trade and travel with pets. Both FAVN (Fluorescent Antibody Neutralization Test) and RFFIT (Rapid Fluorescent Focus Inhibition Test) are approved for determination of the titre of virus neutralizing antibodies.
ELISAs provide a rapid serological test that avoids the requirement to handle live rabies virus. Those tests detect antibodies that can specifically bind to rabies virus antigens, primarily the rabies virus glycoprotein and nucleoprotein.
For more information see the rabies surveillance blueprint and the OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals